Buzzing. Biting. Itchiness. Mosquitoes can be annoying but can also be a carrier of many diseases, including malaria. According to the Centers for Disease Control and Prevention, 198 million cases of malaria were estimated to have occurred and 500,000 died of the disease worldwide in 2013.
Most of these cases are caused by malaria parasites transmitted by mosquitoes of the genus Anopheles. To spread the malarial parasite, the female mosquito first feeds on the blood of an infected human. Inside the mosquito, the parasites multiply, mature and eventually move into the salivary glands. From there, some parasites exit via the salivary duct to be injected into another person during a subsequent blood feeding.
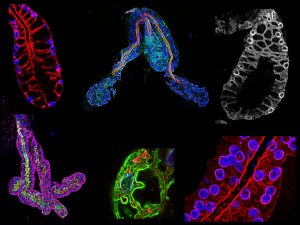
Image courtesy of Michael Wells
A number of strategies have been employed for disease control, including mosquito nets, biological predator introduction, insect repellants and anti-malarial medications. However, there is evidence of mosquitoes developing resistance to such insecticides along with unwanted ecosystem disturbances as a result of some of the biological introductions. For these reasons, new approaches are being developed to better deal with the issue on multiple levels.
Michael Wells, a postdoctoral fellow, and Deborah Andrew, a faculty member in the Department of Cell Biology, recently published a study in which they developed a fluorescence staining method to visualize cellular architectural features in the salivary glands of Anopheles stephensi, a major vector species of malaria. The method used in this study confirms many findings revealed previously in the literature with electron microscopy, but it also allows for more samples to be analyzed in greater detail in regard to how the cellular components are organized.
The study includes comparisons of salivary gland structure between Anopheles stephensi (mosquito) and Drosophila melanogaster (fruit fly), finding many conserved features even though the function and general shape of the salivary gland in these two insects is different. On a basic science level, this experimental approach could be used to address many other types of questions about cellular structure.
To illustrate the findings of the paper, Wells shows me some images of salivary glands of male and female Anopheles stephensi mosquitoes. Even to my untrained eye, the difference is striking. The structure of the female gland is more elaborate and has more lobes than that of the male. The end of the thin ducts — through which the malaria parasites travel — look “open” in the female but are “fused” in the male. The female mosquitoes are the parasite vectors, so this cellular structure makes sense in regard to transmission.
One could imagine using genetic manipulation to change these duct structures as an anti-malarial strategy, such as making the female ducts structurally more similar to that of the male. Within the images, Wells points out some female samples, which include single “fused” ducts, are similar to those found in males. Wells says they have focused on these structures because “the duct is actually small enough that parasites travel in single file to get through so any perturbation of the duct could be very limiting for parasite transmission.”
In addition to the contributions to basic science, results like these have broader public health implications. And, of course, there are larger ecologic questions to consider. Genetic modifications that would disrupt salivary gland function may eradicate mosquito populations and negatively impact ecosystems. But instead, what if it were possible to develop a mutant mosquito that is only deficient in its ability to transmit parasites but is otherwise capable of normal function? Understanding the structure of these systems is one of the first steps toward realizing these kinds of possibilities.
Ugh. No fun!Hopefully it is just idiopathic, but it woludn't hurt to go to a clinic to make sure you don't have a chronic problem. Sjf6gren's syndrome can be pretty serious.
Comments are closed.